Synaptic Pruning and the Dual-Pathway Model of Autism
Genetic Sensitivity, Trauma Adaptation, and the Ecology of Connection
Abstract
Autism Spectrum Disorder (ASD) has long been conceptualized as a neurodevelopmental disorder characterized by differences in communication, social reciprocity, and sensory processing. However, emerging research in neurobiology, trauma studies, and social neuroscience challenges the deficit-based paradigm by reframing autism as an adaptive signal within the broader ecology of human connection. This essay integrates findings on synaptic pruning abnormalities in ASD with a Dual-Pathway Model that distinguishes between genetically inherited sensitivity and trauma-induced adaptation. Drawing upon polyvagal theory and attachment research, it argues that atypical synaptic pruning reflects the body’s negotiation between connection and protection, two fundamental imperatives of the human nervous system. Early-onset autism manifests as inherited fidelity to coherence, while trauma-activated autism represents the body’s response to relational fragmentation. Together, they reveal autism not as pathology but as a physiological communication about the state of relational safety within a culture increasingly disconnected from itself. The essay concludes that the rising prevalence of autism reflects collective dysregulation rather than genetic decay, calling for an ecological model of care that restores relational attunement across individual, familial, and societal systems.
Introduction
Autism Spectrum Disorder (ASD) has historically been defined through behavioral and cognitive differences that deviate from socially normative expectations. The biomedical model, grounded in deficit-based frameworks, has often failed to capture autism’s adaptive and communicative dimensions. As global prevalence rates continue to rise (Zablotsky et al., 2023; Solmi et al., 2022), it becomes imperative to reconsider whether autism represents individual pathology or a collective signal of environmental and relational disarray.
Recent developments in neuroscience and developmental psychology have revealed that many core features of autism, including sensory hypersensitivity, intense focus, and resistance to social ambiguity, are linked to differences in synaptic pruning and neural connectivity. Synaptic pruning, the process by which the brain refines its connections during early development, is central to understanding how individuals interpret and respond to their environments (Tang et al., 2014). Abnormal pruning has been implicated in autism, leading to either excessive or inefficient synaptic networks (Hutsler & Zhang, 2010).
Trauma research demonstrates that chronic stress and attachment disruptions produce parallel alterations in neural connectivity and social engagement (Danese & McEwen, 2012; van der Werff et al., 2013). Integrating these findings suggests that autism may arise from two convergent pathways: a genetic sensitivity pathway reflecting inherited neurobiological attunement to coherence, and an environmental trauma-adaptation pathway reflecting learned hypersensitivity after safety has failed.
The Dual-Pathway Model of Autism frames these biological and environmental mechanisms as complementary rather than opposing. It reinterprets autism as the body’s effort to preserve truth and coherence in the face of relational incongruence. By situating synaptic pruning within this framework, this essay bridges cellular neuroscience and social ecology, demonstrating how the structure of neural networks mirrors the structure of human relationships.
Theoretical Background: Synaptic Pruning and Neural Connectivity
During early brain development, the human brain undergoes massive synaptogenesis, forming far more synaptic connections than it will ultimately retain. Synaptic pruning selectively eliminates weaker or redundant synapses, refining communication efficiency (Huttenlocher & Dabholkar, 1997). This process is guided by experience, attachment, and environmental feedback. Neurons that repeatedly fire together during meaningful relational interactions are preserved, while those lacking coordinated input are pruned away (Katz & Shatz, 1996).
In autism, this pruning process appears altered. Tang et al. (2014) reported that children with ASD exhibited up to 50% more synapses in cortical regions than typically developing peers, suggesting impaired autophagy, a cellular recycling mechanism that governs pruning. Related studies implicate dysregulation in the mTOR signaling pathway, which normally balances synaptic growth and elimination. Overactivation of mTOR suppresses autophagy, leading to synaptic overgrowth and impaired refinement.
Microglia, the immune cells of the brain, also play a crucial role in synaptic pruning by tagging inactive synapses for removal via the complement cascade (C1q, C3, C4). Postmortem and animal studies indicate atypical microglial activity in ASD (Sekar et al., 2016). These cellular anomalies align with observed behavioral traits, including heightened sensory processing, difficulty filtering stimuli, and local hyperconnectivity.
The pruning process reflects a neural negotiation between precision and protection. When pruning functions optimally, it maintains efficient, attuned communication networks. When disrupted, it creates excessive sensitivity or withdrawal, two hallmarks of autistic and trauma-affected nervous systems. This biological dynamic resonates with polyvagal theory’s distinction between states of safety (ventral vagal), defense (sympathetic), and shutdown (dorsal vagal) (Porges, 2011).
The Dual-Pathway Model of Autism
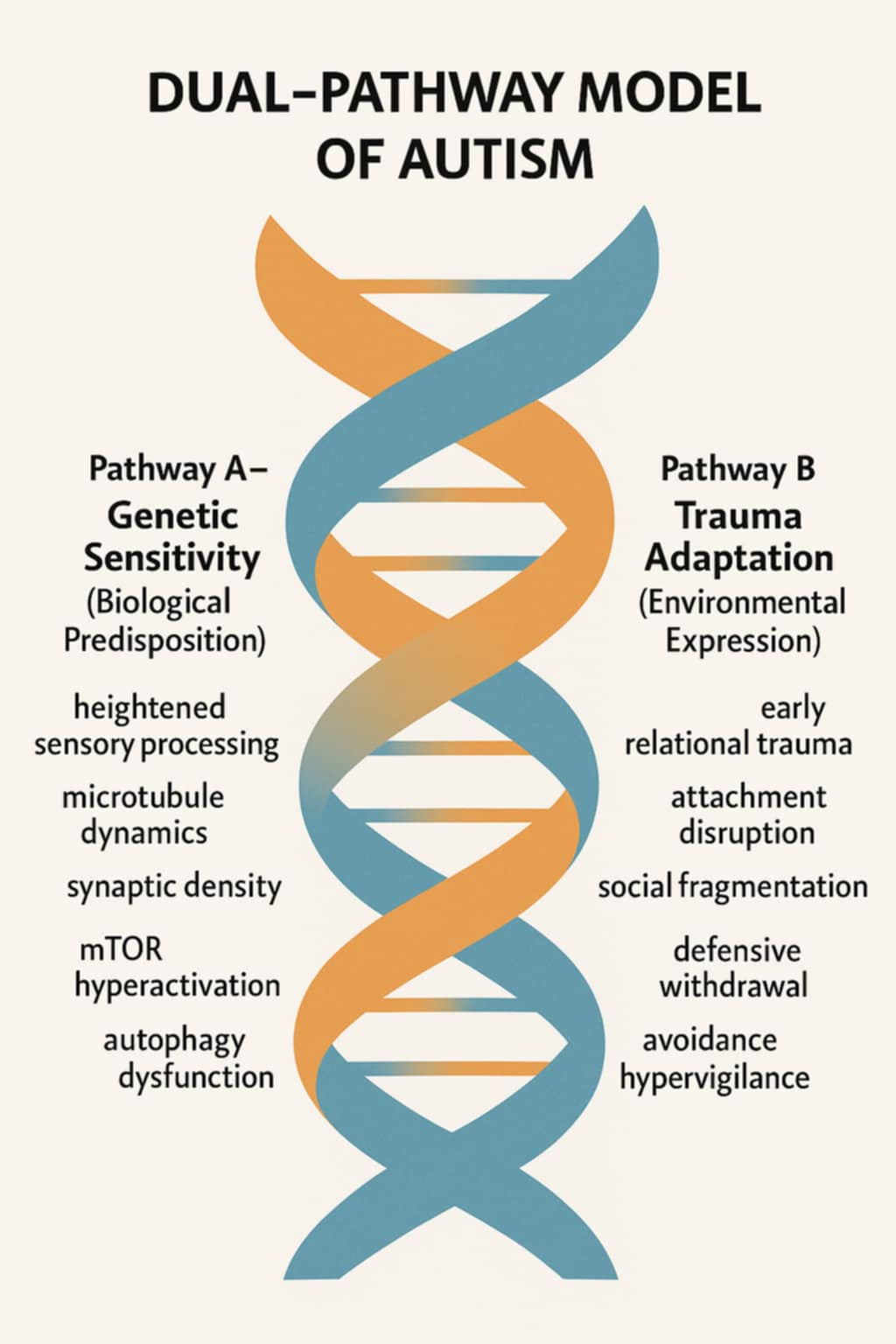
The Dual-Pathway Model posits two distinct but interrelated routes through which autism emerges: (1) genetic sensitivity to relational discord and (2) environmental sensitization through trauma. Both pathways use the same physiological architecture, synaptic pruning, to express divergent histories of connection.
Genetic Sensitivity Pathway
Large-scale genomic studies identify autism-associated variants in genes regulating synaptic function and scaffolding, including SHANK3, CNTNAP2, and NLGN4. These genes influence how neurons form and maintain synapses, determining the threshold for pruning. Rather than producing deficits, these variants confer heightened fidelity to coherence, meaning that the autistic brain preserves detail and pattern integrity at the cost of social smoothing (Dawson et al., 2004).
This biological precision may represent an evolutionary specialization. Individuals with enhanced sensitivity to inconsistency could serve as stabilizers within social groups. From this perspective, early-onset autism is not maladaptive but ecologically corrective, ensuring that social systems maintain honesty, predictability, and safety. However, when such sensitivity meets environments organized around coercion or inauthenticity, industrialized cultures that reward masking and suppress emotion, the same trait becomes a liability.
Inefficient pruning symbolizes a nervous system that refuses to discard information prematurely. This resistance mirrors a psychological and moral orientation toward truth, aligning with the observation that many autistic individuals display deep intolerance for hypocrisy and contradiction. Their neurological structure literally encodes a commitment to coherence.
Trauma Adaptation Pathway
Conversely, late-onset or trauma-activated autism arises when environmental stress chronically dysregulates the nervous system. Prolonged activation of the hypothalamic–pituitary–adrenal (HPA) axis increases cortisol and inflammatory cytokines, disrupting synaptic maintenance and promoting maladaptive pruning (Danese & McEwen, 2012).
Epigenetic studies demonstrate that adverse experiences methylate key genes regulating stress and social behavior, particularly OXTR (oxytocin receptor) and NR3C1 (glucocorticoid receptor), reducing oxytocin signaling and altering limbic-prefrontal connectivity (McGowan et al., 2009). The resulting neuroarchitecture mirrors autistic features such as sensory overload, reduced eye contact, and emotional blunting.
Trauma-based dysregulation represents the learned expression of the same sensitivity inherited in early-onset autism. The difference lies in causation. One arises from genetic fidelity to coherence, the other from environmental betrayal of safety. Both express the same relational law: when attunement fails, the nervous system reorganizes toward self-preservation.
Synaptic Pruning as the Bridge Between Pathways
Synaptic pruning serves as the biological hinge uniting these two pathways. In early-onset autism, reduced pruning maintains an expanded web of neural connections, representing connection without safety, a brain open to the world but overwhelmed by its inconsistency. In trauma-induced autism, excessive or chaotic pruning represents safety without connection, a defensive narrowing of perception after trust has been violated.
Pruning embodies the dialectic between connection and protection. Both extremes emerge when the nervous system lacks reliable co-regulation. The healthy balance of pruning depends on predictable relational feedback, precisely what secure attachment provides (Ainsworth, 1978; Bowlby, 1988).
When early caregivers model emotional consistency, microglial and mTOR systems calibrate accordingly, pruning only redundant connections. When caregivers are inconsistent or frightening, the system learns that no pattern is safe; pruning becomes erratic or incomplete. Over time, the brain’s wiring becomes a map of relational history, a mirror of the ecology of connection in which it developed.
The Ecology of Connection
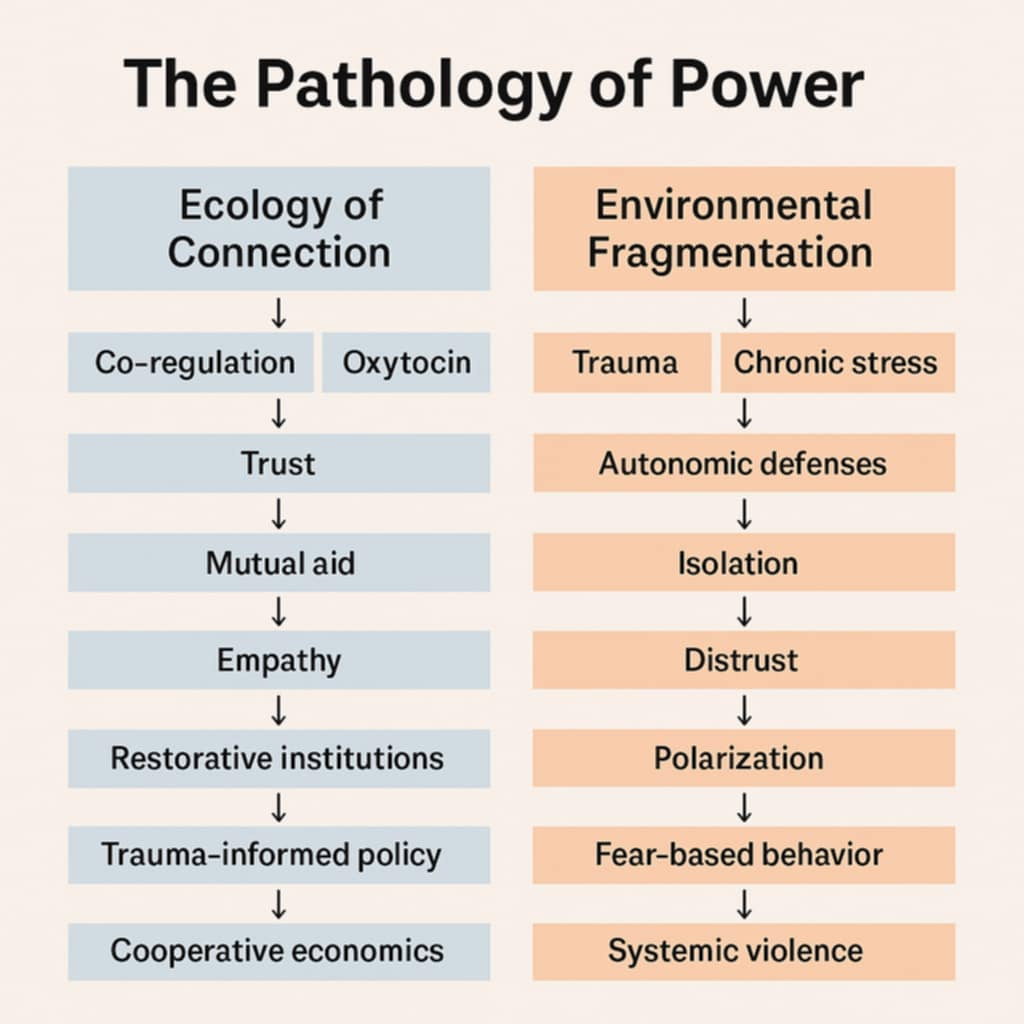
Human beings are not isolated cognitive processors but nodes within a living network of relational energy. Ecologist Suzanne Simard’s (2021) discovery of the “Wood Wide Web” demonstrates that forest health depends on communication and nutrient-sharing among trees via mycorrhizal networks. Likewise, interpersonal neurobiology (Siegel, 1999) reveals that emotional regulation depends on relational feedback loops between human nervous systems.
Synaptic pruning functions analogously: neurons communicate and reorganize based on feedback from other neurons. This neural ecology mirrors the social ecology of attachment and trust. In secure environments, both neurons and people learn that connection is safe, fostering integration and efficiency. In unsafe environments, both retract communication to survive, creating fragmentation and inefficiency.
Autism, viewed through this ecological lens, is the body’s commentary on the relational field. Rising prevalence in industrialized societies parallels increasing social isolation, economic inequality, and sensory overload (Solmi et al., 2022). The autistic brain’s refusal to prune, to discard connection too easily, may therefore represent a biological insistence on maintaining integrity in a culture addicted to fragmentation.
Lori Hogenkamp’s Evolutionary Stress Framework (ESF) deepens this ecological view by framing stress not as pathology but as an adaptive language of communication within complex biological and social systems. Just as trees in Suzanne Simard’s “Wood Wide Web” regulate their ecosystems through dynamic exchanges of nutrients and information, ESF suggests that human neurotypes and stress responses function as adaptive calibrations within an ever-evolving relational network. When environmental or social conditions become chronically dysregulating, through isolation, inequality, or sensory overload, then stress signals proliferate, much like an ecosystem under strain. From this perspective, autism’s altered patterns of synaptic pruning and sensory regulation are not malfunctions but evolutionary adaptations to relational environments marked by fragmentation. The autistic nervous system’s heightened sensitivity and tendency toward connectivity echo ESF’s premise that biology strives to preserve coherence and relational integrity even under systemic stress, making autism a vital expression of the collective body’s attempt to restore ecological balance within an overstressed civilization.
Polyvagal Theory and the Biology of Safety
Polyvagal theory provides the physiological map that links pruning, attachment, and autism. According to Porges (2011), the nervous system organizes behavior around neuroception, the implicit detection of safety or threat. When safety cues predominate, the ventral vagal complex fosters social engagement, curiosity, and learning. When threat dominates, the sympathetic system mobilizes defense; when overwhelm persists, the dorsal vagal system induces shutdown.
Synaptic pruning depends on ventral vagal dominance. The brain prunes efficiently when safety is consistent. Chronic activation of defensive circuits disrupts pruning, leading either to excessive connectivity, as in hypervigilant states, or underconnectivity, as in dissociative shutdown. Both patterns are observed in autism and trauma-related conditions.
This convergence explains why many autistic individuals exhibit oscillations between hyperarousal and hypoarousal, between overstimulation and withdrawal. Their nervous systems track environmental safety with extraordinary precision. The autistic “meltdown” or shutdown is not misbehavior but a polyvagal response to perceived incoherence.
Discussion: Autism as Communication
Integrating synaptic pruning research with the Dual-Pathway Model reframes autism as communication rather than disorder. The autistic nervous system expresses, at the biological level, the same principle articulated by attachment theory: the need for safety through connection. Genetic sensitivity preserves the ability to detect incoherence, and trauma adaptation exposes the cost of relational failure. Both are adaptive attempts to restore equilibrium.
The convergence of these findings supports a paradigm shift. Autism is not the malfunction of a disordered brain but the manifestation of an attuned organism within a dysregulated society. The body, through its synapses, speaks the truth of its ecology. Rising autism prevalence thus signals not evolutionary decline but collective emotional disorganization, a symptom of modernity’s chronic threat states and relational fragmentation.
Recognizing autism as relational feedback invites ethical transformation. Treatment shifts from normalization to co-regulation; policy shifts from behavioral compliance to environmental attunement. Therapeutic interventions should therefore prioritize relational safety, sensory integrity, and authentic communication rather than suppression of difference.
Conclusion
Synaptic pruning provides the biological foundation for understanding the Dual-Pathway Model of Autism. Both early-onset and trauma-activated autism emerge from disruptions in the nervous system’s negotiation between connection and protection. Genetic variants that reduce pruning preserve coherence at the cost of overwhelm; trauma-induced dysregulation that distorts pruning protects at the cost of openness. Together, they articulate the same truth, that the human organism cannot separate its neural health from the quality of its relationships.
Autism, in this light, is the mirror of our collective nervous system, a signal that modern societies must restore relational safety at every scale. Healing lies in building environments that allow the nervous system to prune, connect, and integrate naturally. Safety, as Stephen Porges reminds us, is not the absence of threat but the presence of connection.
Key Research Links:
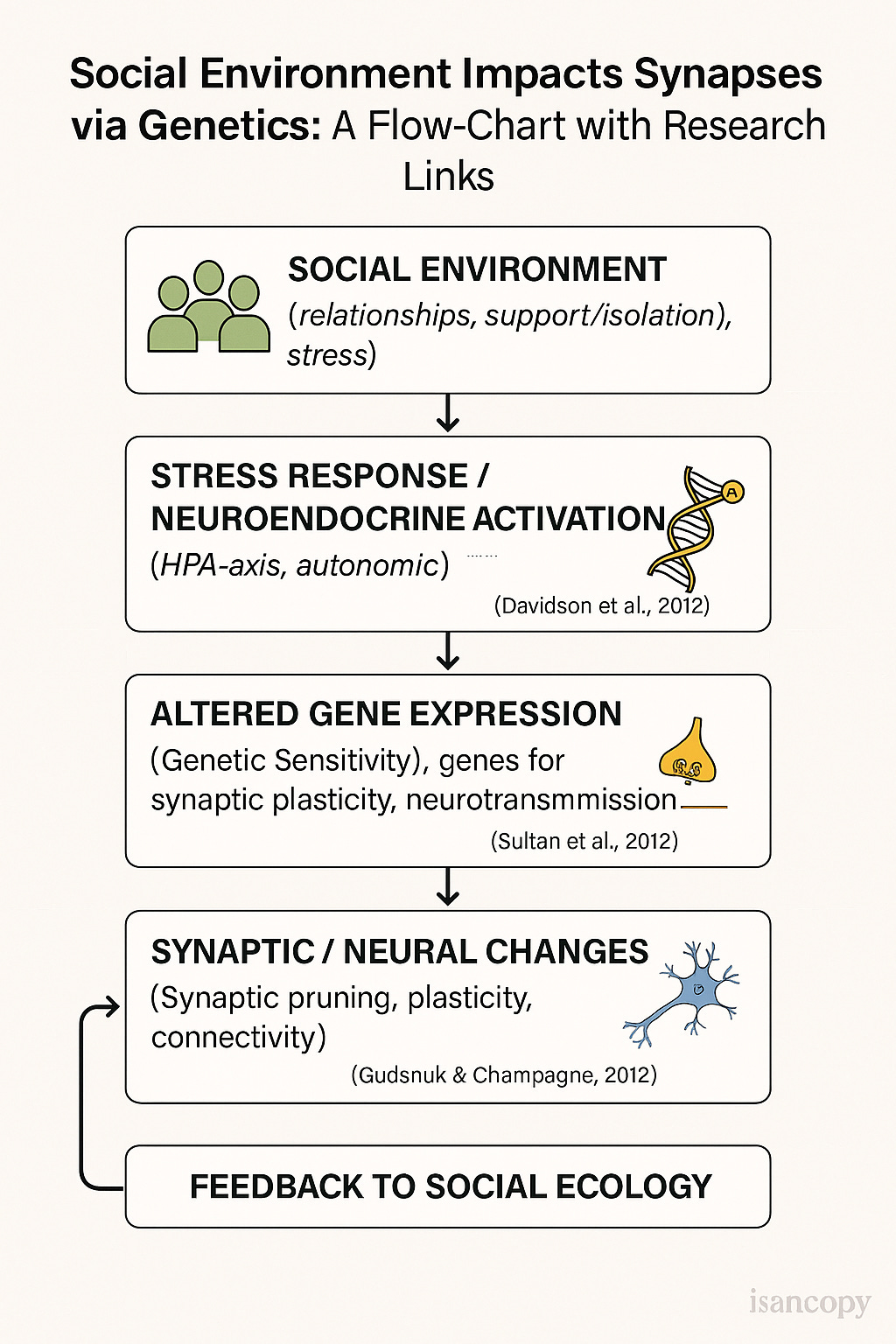
Social influences on neuroplasticity: the review by Richard J. Davidson et al., covers how social contexts shape brain structure/function. PMC
How social environment affects gene expression: Shinichi Toyokawa et al., “How does the social environment ‘get into the mind’?” ScienceDirect
Epigenetic modifications and brain plasticity: review by Fatiha A. Sultan et al., on epigenetic mechanisms in memory and synaptic function. PMC
Epigenetic influence of stress/social environment: Kevin Gudsnuk et al., “Epigenetic Influence of Stress and the Social Environment.” PMC
Recent review on epigenetic modifications & neuroplasticity (early life stress to synaptic change): Bianca M. Benatti et al. “Epigenetic Modifications and Neuroplasticity in the Pathogenesis of Depression.” MDPI
Brain plasticity & epigenetics across development: C. J. Peña et al., “Epigenetic regulation of brain development, plasticity, and plasticity.” Nature
References
Ainsworth, M. D. S. (1978). Patterns of attachment: A psychological study of the strange situation. Erlbaum.
Benatti, B. M., Lucchi, C., Valente, M., & Dell’Osso, B. (2023). Epigenetic modifications and neuroplasticity in the pathogenesis of depression. Behavioral Sciences, 13(10), 882. https://www.mdpi.com/2076-328X/13/10/882
Bowlby, J. (1988). A secure base: Parent-child attachment and healthy human development. Basic Books.
Cozolino, L. (2014). The neuroscience of human relationships: Attachment and the developing social brain (2nd ed.). W. W. Norton & Company.
Danese, A., & McEwen, B. S. (2012). Adverse childhood experiences, allostasis, allostatic load, and age-related disease. Physiology & Behavior, 106(1), 29–39. https://doi.org/10.1016/j.physbeh.2011.08.019
Davidson, R. J., McEwen, B. S., & Sapolsky, R. M. (2012). Social influences on neuroplasticity: Stress, social support, and brain function. Proceedings of the National Academy of Sciences. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3491815/
Dawson, G., Toth, K., Abbott, R., Osterling, J., Munson, J., & Estes, A. (2004). Early social attention impairments in autism. Developmental Psychology, 40(2), 271–283. https://doi.org/10.1037/0012-1649.40.2.271
Feldman, R. (2017). The neurobiology of human attachments. Trends in Cognitive Sciences, 21(2), 80–99.
Frontiers in Cellular Neuroscience. (2020). Dynamics of neurosocial plasticity: How social interaction shapes neural networks. Frontiers in Cellular Neuroscience, 14, Article 271. https://www.frontiersin.org/journals/cellular-neuroscience/articles/10.3389/fncel.2020.00271/full
Gudsnuk, K., & Champagne, F. A. (2012). Epigenetic influence of stress and the social environment. Frontiers in Neuroendocrinology, 33(2), 109–118. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4021821/
Hogenkamp, L. (2023). The Evolutionary Stress Framework (ESF): Understanding stress as an adaptive system of regulation and communication. Center for Adaptive Stress. https://ndstress.wordpress.com/esf-2/
Hutsler, J. J., & Zhang, H. (2010). Increased dendritic spine densities on cortical projection neurons in autism spectrum disorders. Brain Research, 1309, 83–94. https://doi.org/10.1016/j.brainres.2009.09.120
Huttenlocher, P. R., & Dabholkar, A. S. (1997). Regional differences in synaptogenesis in human cerebral cortex. Journal of Comparative Neurology, 387(2), 167–178. https://doi.org/10.1002/(SICI)1096-9861(19971020)387:2<167::AID-CNE1>3.0.CO;2-Z
Katz, L. C., & Shatz, C. J. (1996). Synaptic activity and the construction of cortical circuits. Science, 274(5290), 1133–1138. https://doi.org/10.1126/science.274.5290.1133
McGowan, P. O., Sasaki, A., D’Alessio, A. C., Dymov, S., Labonté, B., & Szyf, M. (2009). Epigenetic regulation of the glucocorticoid receptor in human brain associates with childhood abuse. Nature Neuroscience, 12(3), 342–348. https://doi.org/10.1038/nn.2270
Peña, C. J., Bagot, R. C., & Nestler, E. J. (2025). Epigenetic regulation of brain development and plasticity. Nature Neuroscience, 28(4), 455–468. https://www.nature.com/articles/s41386-025-02179-z
Porges, S. W. (2011). The polyvagal theory: Neurophysiological foundations of emotions, attachment, communication, and self-regulation. W. W. Norton.
Schore, A. N. (2012). The science of the art of psychotherapy. W. W. Norton & Company.
Sekar, A., Bialas, A. R., de Rivera, H., Davis, A., Hammond, T. R., Kamitaki, N., et al. (2016). Schizophrenia risk from complex variation of complement component 4. Nature, 530(7589), 177–183. https://doi.org/10.1038/nature16549
Siegel, D. J. (1999). The developing mind: How relationships and the brain interact to shape who we are. Guilford Press.
Simard, S. (2021). Finding the mother tree: Discovering the wisdom of the forest. Alfred A. Knopf.
Solmi, M., Song, M., Cortese, S., Zheng, P., Polcwiartek, C., Correll, C. U., & Carvalho, A. F. (2022). Global burden of autism spectrum disorder: Trends, causes, and future directions. Molecular Psychiatry, 27(1), 1–14. https://doi.org/10.1038/s41380-022-01630-7
Sultan, F. A., & Day, J. J. (2012). Epigenetic mechanisms in memory and synaptic function. Molecular Psychiatry, 17(6), 590–602. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3350307/
Tang, G., Gudsnuk, K., Kuo, S. H., Cotrina, M. L., Rosoklija, G., & Sulzer, D. (2014). Loss of mTOR-dependent macroautophagy causes autistic-like synaptic pruning deficits. Neuron, 83(5), 1131–1143. https://doi.org/10.1016/j.neuron.2014.07.040
Toyokawa, S., Uddin, M., Koenen, K. C., & Galea, S. (2012). How does the social environment “get into the mind”? Epigenetics at the intersection of social and psychiatric epidemiology. Social Science & Medicine, 74(1), 67–74. https://www.sciencedirect.com/science/article/abs/pii/S0277953611006332
van der Werff, S. J., Pannekoek, J. N., Veer, I. M., van Tol, M. J., Aleman, A., & van der Weert, H. C. (2013). Resting-state functional connectivity in adults with childhood emotional maltreatment. Biological Psychiatry, 73(8), 614–621. https://pubmed.ncbi.nlm.nih.gov/23254143/
Zablotsky, B., Black, L. I., Maenner, M. J., & Schieve, L. A. (2023). Prevalence and trends of autism spectrum disorder among children aged 8 years. MMWR Surveillance Summaries, 72(2), 1–14. https://doi.org/10.15585/mmwr.ss7202a1